
Mendeleev countered this somewhat in his book The Principles of Chemistry in 1891: John Newlands took issue with the credit that Mendeleev was given for the periodic table in his book ‘On the Discovery of the Periodic Law’ in 1884, going so far as to quote Mendeleev as supporting him: "It is possible that Newlands has prior to me enunciated something similar to the periodic law.". Atomic numbers were not yet known, but the atomic weights worked well enough for ordering most of the elements, and Mendeleev was able to accurately predict the properties of missing elements. He also thought to occasionally ignore the order suggested by the atomic weights and to switch adjacent elements where they could be better classified into chemical families. What made his version successful was the fact that he left gaps where there appeared to be an element missing that had not yet been discovered. Mendeleev’s table listed elements in rows or columns in order of atomic weight, starting a new row or column when the characteristics of the elements began to repeat. In fact, he was ill at the time so his colleague Nikolai Menschutkin made the announcement on his behalf – on 6 March at a meeting of the Russian Chemical Society. The records show that Mendeleev discovered the periodic system on 17 February 1869. Due to the seemingly arbitrary nature of his theory and because the Society had a rule of not publishing purely theoretical papers, they declined to publish his work. In addition, flaws in his theory led to him having to make changes to the order of some elements himself as some were occupying the same spaces. When Newlands gave a lecture to the Chemical Society on 1 March 1866 to announce his discovery, he was somewhat derided when he compared the repetition of elements at intervals of eight with a musical octave. Odling’s table was remarkably similar to the one that Mendeleev had devised as a first attempt and actually, Odling had been able to overcome some issues that Mendeleev had not.īetween 18, British chemist John Newlands wrote a series of papers that went on to explain his 'Law of Octaves' having noticed similarities between elements with atomic weights that differed by seven. Meyer never disputed Mendeleev's priority and later stated that he lacked sufficient courage to have gone on to predict the existence of undiscovered elements.Two years later, Julius Lothar Meyer and William Odling independently published their own versions of a periodic table. Meyer’s table was comprised of 44 elements arranged by valency Odling’s version arranged 57 elements by their atomic weight. Meyer did not publish his table until 1870 so he was preempted by Dmitri Mendeleev, who had published his periodic table in 1869. 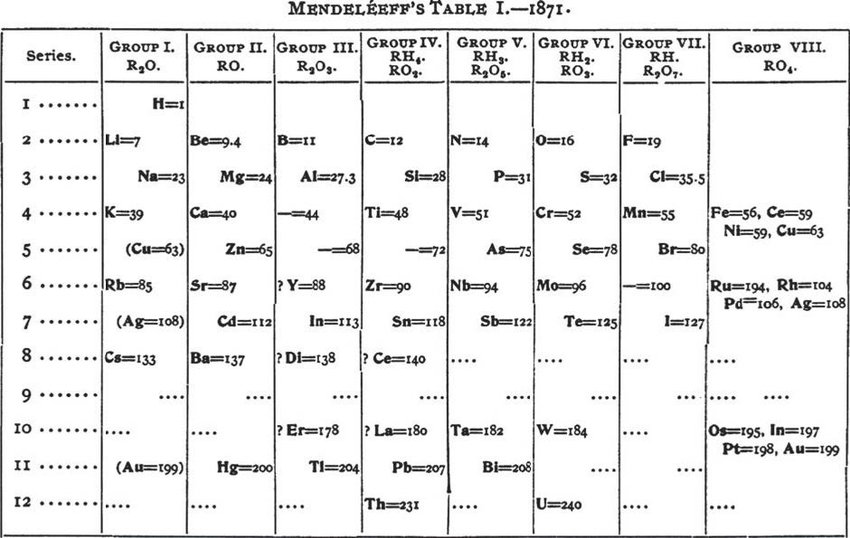
He noticed that elements with similar chemical properties occur at comparable points on the different peaks e.g., the alkali metals all occur at the tops of the peaks. He found clear signs of periodicity, the graph consisting of a series of four sharp peaks. Meyer plotted the values of a certain physical property, atomic volume, against atomic weight. In writing his textbook it had occurred to him that the properties of an element seem to depend on its atomic weight. He was much impressed by Stanislao Cannizzaro, expounding his work in his book Die modernen Theorien der Chemie (1864 Modern Chemical Theory). Meyer is best remembered for his early work on the periodic table. He became professor of chemistry at Karlsruhe in 1868 where he stayed until he moved to the chair at Tübingen (1876–95). At first his interests were physiological but he slowly moved into chemistry. He qualified in medicine himself in 1854 after studying at Zurich and Würzburg and gained his PhD from the University of Breslau in 1858. Meyer was the son of a doctor from Varel in Germany.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
